Mens Sana Monogr. 2012 Jan-Dec; 10(1): 13–19. doi: 10.4103/0973-1229.91298
PMCID:PMC3353592
Abstract
Chlorpromazine efficacy in schizophrenia was observed 60 years ago. Advances in pharmacotherapy of this disorder have been modest with effectiveness still limited to the psychosis psychopathology and mechanism still dependent on dopamine antagonism. While a look backward may generate pessimism, future discovery may be far more robust. The near future will see significant changes in paradigms applied in discovery. Rather than viewing schizophrenia as a disease entity represented by psychosis, the construct will be deconstructed into component psychopathology domains. Each domain will represent a clinical target for aetiologic and therapeutic discovery. Research on pathophysiology will shift to the neural circuit level in relation to specific behavioural constructs. Progress at the molecular, genetic, cellular and network levels will be more robust. The behavioural paradigm will map on to the deconstructed clinical paradigm and in the process discovery will cut across current classification boundaries.
Keywords: Discovery, DSM-5, Paradigms, Pharmacotherapy, Psychopathology, Schizophrenia
Introduction: Modest Past but Bright Future
Andrade and colleagues (this issue[2]) see a modest past and a bleak future for psychopharmacology of schizophrenia. I generally share their view of the past and will note several reasons that have minimized progress. However, I think the future will be based on new paradigms that will alter the nature of discovery and offer promise for novel therapeutic mechanisms relating to unmet treatment needs in schizophrenia including primary and secondary prevention.
From Chlorpromazine to Today
Progress in drug discovery for schizophrenia in the 60 years since chlorpromazine was discovered as a treatment for psychosis has been disappointing. Andrade and colleagues[2] describe many of the problems. I have critiqued this issue elsewhere (Carpenter and Koenig 2008,[7] Carpenter 2004[6]). The rationale for development of drugs for schizophrenia depends on understanding the molecular pathophysiology in order to identify molecular targets for compound development. This challenge has not been met, in part because schizophrenia is a heterogeneous syndrome and the human brain has been difficult to access for molecular studies at the tissue level. Neuroimaging techniques can identify involved pathways, but do not address what has caused aberrant structure or function. Genetic studies identify candidate genes, but validation is difficult and, in any case, particular genetic contributions make a very small contribution to manifest illness and associated genetic findings will vary from case to case.
Another major reason for the shortfall in discovery relates to treating schizophrenia as a disease defined by psychosis. This has led to antipsychotic drugs approved for the treatment of schizophrenia but limited in the breath of therapeutic effect with cognition impairment and negative symptoms being the two leading unmet therapeutic needs (Buchanan et al.,2005[5] and Kirkpatrick et al.,2006[11]). Commercial considerations may have also played a major role. Methods for developing an antipsychotic compound have been well established including rodent models that predict the human effect and a known mechanism of action—dopamine D2 antagonism. Based on this model scores of drugs are now on the market, but without novel mechanism or substantial advance in efficacy. An image of second generation antipsychotic drugs being superior to first generation drugs including efficacy for negative symptoms and cognition was created, but is not substantiated with data with the exception of clozapine superiority for treating the psychotic component of the disorder in patients inadequately responsive to other dopamine antagonists. The cost of treatment with second generation antipsychotic drugs created a strong incentive to produce “me-too” drugs.
Clinicians now have a wide range of adverse effects to guide selection for individual patients, but still only one drug with established superiority. Major pharmaceutical companies are now abandoning the search for new drugs for schizophrenia. Pessimism is understandable, but is the field ready to overcome pessimism regarding drug discovery for persons with schizophrenia?
My answer is yes, based on the following considerations.
A Paradigm Shift
A significant shift in paradigm is taking place that will change the target for drug development from schizophrenia to domains of pathology. These domains will cross diagnostic class boundaries. First advocated as clinical therapeutic targets in 1974 (Strauss et al.,1974[15]) and reinforced with the work of Peralta and Cuesta 2001,[14] a series of psychopathology domains is being considered for the psychotic disorders in DSM-5. In addition to diagnostic class, the domains represent the key symptom areas for therapeutic discovery (DSM-V[9]).
The U.S. Food and Drug Administration has agreed on cognition and negative symptoms as potential indications for drug development within schizophrenia (Buchanan et al.,2005[5]; Kirkpatrick et al.,2006[11]). They will be challenged to consider the implications of psychopathology domains crossing diagnostic boundaries.
The NIMH Research Domains Criteria (Cuthbert and Insel 2010[8]) initiative is pushing this paradigm change further calling for the identification of clinical problems at the level of behavioural construct and involved neural circuit. This will provide a more robust design for investigating molecular, cellular and network pathophysiology. Clinical studies will be challenged to relate symptom constructs to specific behavioural constructs.
Secondary Prevention
The past 20 years has produced substantial validation of early identification of young people who have developed a disorder associated with mild manifestations of psychotic-like symptoms. Disorder is confirmed by distress, disability and dysfunction and help seeking. But progression to a full psychotic illness is uncertain and may be influenced by early intervention. There may be a critical point of intervention that truly alters the longer term course. If so, this will be a very significant example of secondary prevention. Although not yet replicated, an exciting report (Amminger et al.,2010[1]) from a random assignment placebo controlled study in this population observed a very substantial advantage for 12 weeks of 3-omega free fatty acid therapy compared to placebo in progression to full psychosis over the next 40 weeks-4% in the experimental treatment and 27% in the placebo group.
Primary Prevention
Finally, the possibility of primary prevention can now be based on extensive information regarding modifiable risk factors. Brown and McGrath (2011);[3] Kirkbride and Jones (2011);[10] McGrath et al.,(2011)[12]; Brown and Patterson (2011)[14] and McGrath and Lawlor (2011)[13]) have recently summarized this evidence. Reduction in cannabis use and assuring adequate vitamin D are but two examples. Improved prenatal care with emphasis on minimizing psychological and biological stress during first and second trimesters and reducing complications in labour and delivery is another.
Conclusions [See Figure 1 Flowchart of Paper]
Figure 1.
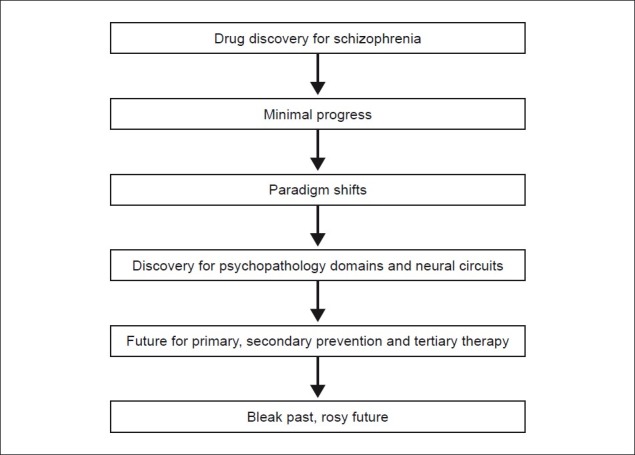
Flowchart of paper
What is necessary now is for the field to cease designing studies at though schizophrenia was a disease entity rather than a syndrome. This will provide more robust psychopathology for investigations and help clarify porous diagnostic boundaries. More precise knowledge of pathophysiology is essential for rationale drug discovery, and animal models that predict therapeutic effects in humans for domains other than psychosis are needed to enable pharmaceutical companies to make early go/no-go decisions on their compounds. Making early detection and intervention (not necessarily with pharmacotherapy) a priority may enhance secondary prevention. Furthermore, evidence on risk factors from epidemiology has prepared the field for applied epidemiology and primary prevention.
Take home message
Andrade and colleagues (this issue[2]) give the following take home message: “Primary prevention in schizophrenia is probably impossible. Secondary prevention approaches have so far met with disappointing results. The focus of drug discovery in schizophrenia lies in the realms of tertiary prevention, when the phenotype manifests, by which time extensive and probably irreversible structural and functional changes have developed. The future of the pharmacotherapy of schizophrenia appears bleak.”
While sharing disappointment in progress to date, my view is far more optimistic. Primary prevention is feasible with substantial evidence on risk factors and interventions are hypothesized (Brown and McGrath 2011[3] ;Kirkbride and Jones[10]; McGrath et al.,2011[12]; Brown and Patterson 2011[4]) and McGrath and Lawlor 2011[13]). We are early in the testing of secondary prevention, but data to date are consistently showing intervention effectiveness with the remarkable possibility of a critical point of intervention altering the long-term course. Regarding tertiary prevention, paradigm shifts have already resulted in redefining therapeutic targets from a clinical perspective that broaden the window of discovery and address the unmet therapeutic needs of persons with schizophrenia.
Questions that this Paper Raises
What are the clinical design issues for clinical trials to test efficacy for negative symptoms and cognitive impairments?
What paradigm shifts are necessary to break the logjam of discovery for pharmacotherapy of schizophrenia?
How can the porous boundaries between current diagnostic classes be addressed in future research?
In therapeutic discovery, how will psychopathology domains such as avolition map on to behavioral constructs such as positive valence?
About the Author

Dr. Carpenter is Director of the Maryland Psychiatric Research Center, Editor-in Chief of the Schizophrenia Bulletin, and chairs the Psychosis Work Group for DSM-5. His research has focused on psychopathology and clinical trials or novel strategies and compounds. Since 1974, he has advocated a new paradigm for investigating etiology, pathophysiology and treatment of schizophrenia based on deconstruction to domains of pathology. Together with colleagues he established Deficit Schizophrenia as a candidate disease entity within the schizophrenia syndrome. His current work involves integrating animal models, human non-clinical models and clinical trials models in the discovery of novel therapeutics for negative symptoms and cognition impairment. His research has been recognized with leading international honorific awards and he is a member of the Institute of Medicine or the National Academies of Science, U.S.A.
Acknowledgments
Support for research received from the U.S. National Institute of Mental Health and VA Capitol Health Care Network (VISN 5) MIRECC.
Footnotes
Conflict of interest: No stock holdings or speaker bureau. During past 12 months served as advisory to Astra Zeneca and Shire Pharmaceuticals Patent (without potential for personal funds):European Regional Patent Number 1487998 (June 6, 2007) “Methods for Diagnosing and Treating Schizophrenia”
Declaration
This is an original editorial neither published nor submitted elsewhere.
CITATION: Carpenter Jr WT. The Future of Schizophrenia Pharmacotherapeutics: Not So Bleak. Mens Sana Monogr 2012; 10: 13-9.
References
1. Amminger GP, Schafer MR, Papageorgiou K, Klier CM, Cotton SM, Harrigan SM, et al. Long-Chain omega-3 Fatty Acids for Indicated Prevention of Psychotic Disorders: A Randomized, Placebo-Controlled Trial. Arch Gen Psychiatry. 2010;67:146–54. doi: 10.1001/archgenpsychiatry.2009.192. PMID: 20124114. [DOI] [PubMed] [Google Scholar]
2. Andrade C, Radhakrishnan R, Fernandes PP. Psychopharmacology of schizophrenia: The future looks bleak. Mens Sana Monogr. 2012;10:4–12. doi: 10.4103/0973-1229.91293. [DOI] [PMC free article] [PubMed] [Google Scholar]
3. Brown AS, McGrath JJ. The prevention of schizophrenia. Schizophr Bull. 2011;37:257–61. doi: 10.1093/schbul/sbq122. PMID: 20980445. [DOI] [PMC free article] [PubMed] [Google Scholar]
4. Brown AS, Patterson PH. Maternal infection and schizophrenia: Implications for prevention. Schizophr Bull. 2011;37:284–90. doi: 10.1093/schbul/sbq146. PMID: 21134972. [DOI] [PMC free article] [PubMed] [Google Scholar]
5. Buchanan RW, Davis M, Goff D, Green MF, Keefe RSE, Leon AC, et al. A summary of the FDA-NIMH-MATRICS workshop on clinical trial design for neurocognitive drugs for schizophrenia. Schizophr Bull. 2005;31:5–19. doi: 10.1093/schbul/sbi020. PMID: 15888422. [DOI] [PubMed] [Google Scholar]
6. Carpenter W.T., Jr Clinical constructs and therapeutic discovery. Schizophr Res. 2004;72:69–73. doi: 10.1016/j.schres.2004.09.003. PMID: 15531408. [DOI] [PubMed] [Google Scholar]
7. Carpenter WT, Koenig JI. The evolution of drug development in schizophrenia: Past issues and future opportunities. Neuropsychopharmacology. 2008;33:2061–71. doi: 10.1038/sj.npp.1301639. PMID: 18046305. [DOI] [PMC free article] [PubMed] [Google Scholar]
8. Cuthbert BN, Insel TR. Toward New Approaches to Psychotic Disorders: The NIMH Research Domain Criteria Project. Schizophr Bull. 2010;36:1061–2. doi: 10.1093/schbul/sbq108. PMID: 20929969. [DOI] [PMC free article] [PubMed] [Google Scholar]
9. DSM-5: The Future of Psychiatric Diagnosis. [Last accessed on 24 Dec 2011]. Available from: http://www.dsm5.org .
10. Kirkbride JB, Jones PB. The prevention of schizophrenia – what can we learn from eco-epidemiology? Schizophr Bull. 2011;37:262–71. doi: 10.1093/schbul/sbq120. PMID: 20974748. [DOI] [PMC free article] [PubMed] [Google Scholar]
11. Kirkpatrick B, Fenton WS, Carpenter WT, Jr, Marder SR. The NIMH-MATRICS consensus statement on negative symptoms. Schizophr Bull. 2006;32:214–9. doi: 10.1093/schbul/sbj053. PMID: 16481659. [DOI] [PMC free article] [PubMed] [Google Scholar]
12. McGrath J, Brown A, St. Clair D. Prevention and schizophrenia – the role of dietary factors. Schizophr Bull. 2011;37:272–83. doi: 10.1093/schbul/sbq121. PMID: 20974747. [DOI] [PMC free article] [PubMed] [Google Scholar]
13. McGrath JJ, Lawlor DA. The search for modifiable risk factors for schizophrenia. Am J Psychiatry. 2011;168:1235–8. doi: 10.1176/appi.ajp.2011.11081300. PMID: 22193665. [DOI] [PubMed] [Google Scholar]
14. Peralta V, Cuesta MJ. How many and which are the psychopathological dimensions in schizophrenia.? Issues influencing their ascertainment. Schizophr Res. 2001;49:269–85. doi: 10.1016/s0920-9964(00)00071-2. PMID: 11356588. [DOI] [PubMed] [Google Scholar]
15. Strauss JS, Carpenter WT, Jr, Bartko JJ. The diagnosis and understanding of schizophrenia. Part III. Speculations on the processes that underlie schizophrenic symptoms and signs. Schizophr Bull. 1974;11(Winter):61–9. doi: 10.1093/schbul/1.11.61. PMID: 4469362. [DOI] [PubMed] [Google Scholar]

